How plant-eaters snag their essential amino acids
Early in evolution, we animals lost the ability to manufacture nine of the 20 building blocks needed to make proteins. Herbivores evolved an impressive array of tricks to ensure their dietary needs are met.
Support sound science and smart stories
Help us make scientific knowledge accessible to all
Donate today
In warm, shallow waters, a spiny, slug-like creature grazes on bacterial sludge on the seafloor, while sponges nearby filter clouds of suspended particles. A shell-encased Odaraia swims past, picking smaller life forms out of the water, while a giant Anomalocaris, with its formidable grasping appendages, prowls for soft-bodied critters. It’s a scene from some 500 million years ago, of oceans teeming with a new kind of life — the up-and-coming kingdom of animals.
Animals, made of many cells and feasting on other life instead of manufacturing their own nourishment from scratch, were an evolutionary hit. But this way of living brought complications. Early in the kingdom’s lineage, animals lost the ability to make nine of the 20 amino acids needed to build proteins.
The loss made sense: “It saves expending energy on making them ourselves when we can get them from the world around us,” says biochemist Benjamin Pickard of the University of Strathclyde in Scotland. But it has also made animals vulnerable to nutrient deficits. Today, scientists are learning how animals have reconfigured their bodies to detect amino acid deficiencies and react to times of scarcity, and the strategies they’ve evolved to ensure they get enough of the right mix.
In all kinds of ways, Pickard says, as ancient animals diversified into predators and herbivores, “we had to adapt our bodies and life strategies … to supply this permanent hunger for essential amino acids.”

The burst in animal diversity that occurred during the Cambrian period 530 million years ago brought some complications: Early during evolution, animals lost the ability to synthesize nine of the 20 amino acids their bodies require, so they must source them from other life forms. They evolved an array of feeding strategies to satisfy that need.
CREDIT: CHRISTIAN JEGOU / SCIENCE SOURCE
The world is our protein
Even when they’re not actively growing, all animals are in constant need of protein. Our bodies are steadily losing proteins or breaking down old and damaged ones. They must be replaced. For that, our cells need amino acids, organic molecules consisting of a nitrogen-containing stub attached to a carbon-studded molecule. Of the 20 different amino acids we need, our cells can synthesize 11 of them; all they require is a carbon source like sugar or fat, and nitrogen, which we can get from virtually any kind of protein we ingest.
Not that we have to manufacture them: The body can also get these “nonessential” amino acids — which include alanine, asparagine and aspartate — from the diet, to save the energy of synthesizing them.
But the “essential” amino acids that the body can’t produce — like tryptophan, methionine, lysine and leucine — we can get only from our diet. Then there are also several “conditionally essential” amino acids like cysteine and arginine that our bodies normally generate in sufficient amounts, except when demand skyrockets during periods of growth, injury or sickness. In such cases, we need a top-up from our diet. In a 2024 paper, Pickard found that the proteins like keratin that make up human nails and hair contain a lot of these conditionally essential amino acids, which could help to explain why hair and nails tend to grow more slowly or look less healthy when we’re ill: We need to use the amino acids for making more critical proteins.
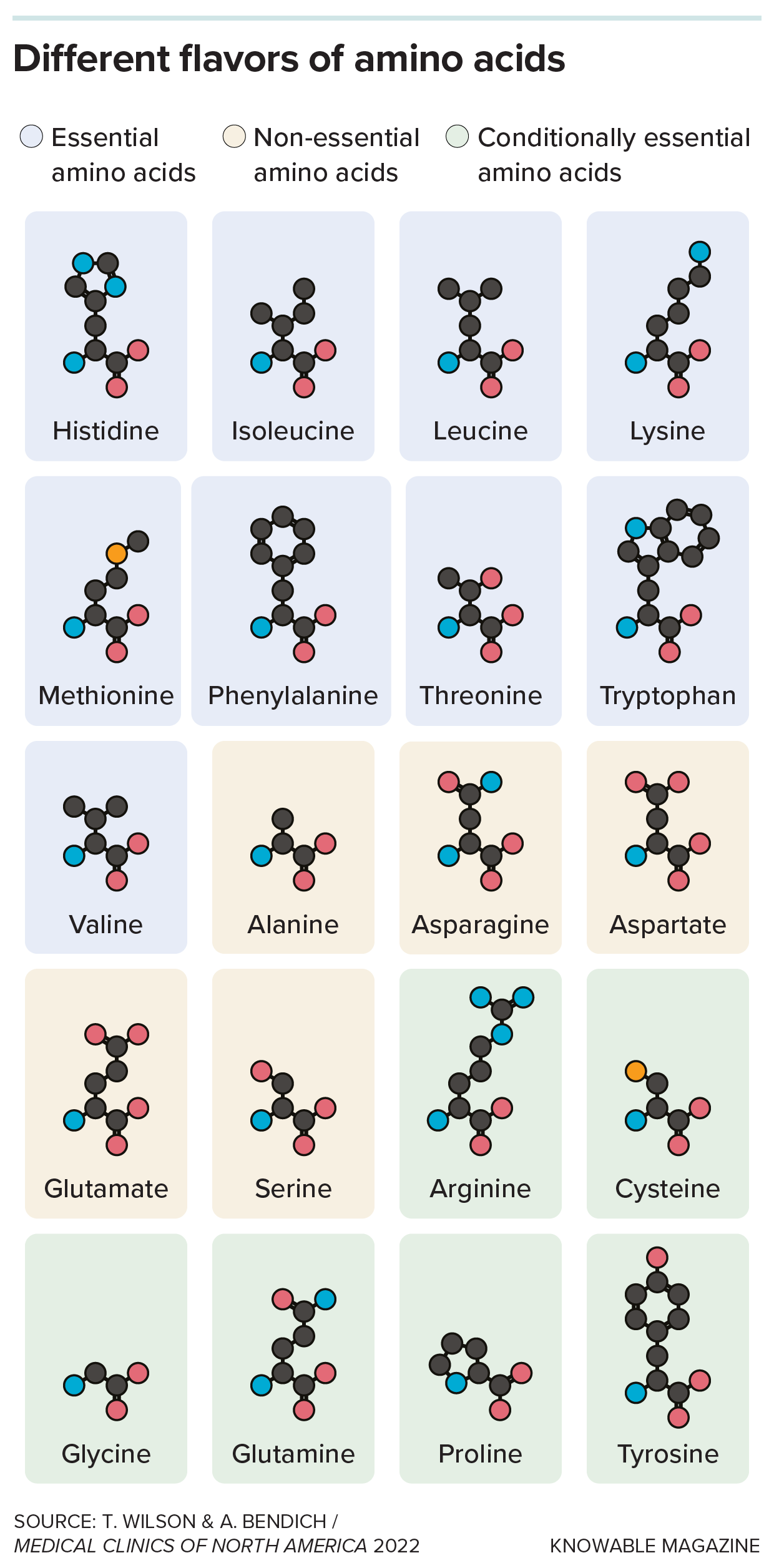
Nine of the amino acids used to build proteins cannot be made by animals; these are termed “essential” amino acids and must be obtained from the diet. Animals can synthesize the remaining, “nonessential,” ones. But some of these — termed “conditionally essential” — must be obtained from the diet during periods of growth, injury or sickness.
Meat provides animals a balanced amino acid profile with all 20 amino acids, but it can be harder to get this from plants. For one thing, specific plant tissues like leaves contain relatively little protein overall. And although soybeans, amaranth, quinoa, potatoes and pistachios are “complete proteins” — they contain sufficient concentrations of all nine essential amino acids — other seeds aren’t.
Almonds and peanuts are low in lysine and methionine; peas and beans lack methionine and tryptophan; rice, wheat and corn are all low in lysine and isoleucine. Chemical ecologist Georg Jander of the Boyce Thompson Institute, a life science research center in New York, speculates that this could be a strategy evolved by plants to deter animals from eating them. “It’s probably less likely that animals will eat your seeds if they’re not nutritionally good for these animals,” he says.
This nutritional inadequacy presents a challenge for herbivorous animals, says animal ecologist Seth Newsome of the University of New Mexico. “It’s really easy to make a wolf, because wolves eat basically what their body looks like. Their diet mirrors their body,” he says. “But it’s really, really hard to make a moose, because a moose’s diet does not.” Herbivores often avoid deficiencies through bacteria in their gut microbiome that digest the cellulose walls of plant cells, releasing extra protein, and synthesize essential amino acids that their host can then use. Many people consume a diversity of protein-rich plant parts that complement each other’s missing amino acids, for instance by pairing beans and rice.
But essential amino acid deficiencies exist in malnourished populations who rely heavily on staple crops like corn; one 2022 diet survey of people in Malawi estimated that 33 percent of households weren’t eating enough lysine. Such deficiencies can have particularly devastating effects in growing children. In developed countries, protein deficiencies are extremely rare, although people on entirely plant-based diets need to take care to get a balanced mix of amino acids; one 2025 study of nearly 200 vegans in New Zealand estimated that about half of participants might not be getting enough lysine and leucine.
When deficiency strikes
Animals have evolved ways of sensing both the overall amount of protein in their meals and the quality. Lab rats, for instance, can tell if the essential amino acid isoleucine is entirely missing from their meal, making them reluctant to eat it. Lysine-deprived rats prefer to drink lysine-containing solutions over other liquids they’re offered. “The capacity to detect nutrients is one of the most fundamental things an organism has to do,” says molecular biologist David Sabatini of the Institute of Organic Chemistry and Biochemistry, based in Boston and Prague in the Czech Republic. “If you don’t detect the absence of nutrients, you basically are like a runaway train that is consuming without bringing things in.”
But how, exactly, is it done? Part of the answer lies in the brain, says neuroscientist Clémence Blouet of the University of Cambridge in the United Kingdom. Scientists have worked out that the brain tracks the quantity and quality of ingested protein, both by reading signals from the gut and by sensing nutrients in the blood. If all is well, it creates feelings of fullness. Curiously, no amino acid has a stronger effect on satiety than leucine: Blouet suspects that it’s because leucine is a good indicator for the quality of a meal — if leucine is present, odds are good that other essential amino acids are, too.
To work out the mechanism, Blouet and her colleagues examined a group of nerve cells in the brains of mice that are known to regulate satiety. They identified a particular protein in the membranes of these cells, called Cav3.1. When leucine binds to Cav3.1, the neurons become more sensitive to a satiety-boosting hormone called GLP-1. This is how leucine, once enough of it is absorbed through our guts into the blood, tells the brain to suppress appetite.
But the brain isn’t the only organ keeping track of amino acids. Many cells — in the gut, muscles and skin — in the bodies of fruit flies, mice and humans have sophisticated machinery to detect deficiencies and adapt accordingly. Specialized proteins sense the cells’ concentrations of essential amino acids like leucine and methionine as well as conditionally essential ones like arginine. Upon detecting a deficiency, they relay signals to a nutrient-sensing hub of proteins, which then prompts a powerful protein called mTORC1 to order a slowdown in protein-making inside the cell, scientists have learned.
And should the cell’s protein factories — the ribosomes — be in the middle of producing a protein but a particular amino acid is missing, they stop in their tracks, leaving behind a half-baked protein that promptly gets degraded by the cell. This, research shows, activates another protein, GCN2, that triggers a slowdown of protein synthesis.
Importantly, these systems also instruct cells to speed up the destruction of old or unnecessary proteins to make amino acids available for making more necessary ones. “Basically, we start eating our own cells,” says biochemist Stefan Bröer of Australian National University, who authored an article on amino acid absorption in the 2023 Annual Review of Nutrition.
Studies hint that creatures also actively reallocate their amino acid resources in times of scarcity. In experiments, stem cell biologist Elaine Fuchs of the Rockefeller University and her colleagues placed mice on diets that lacked serine — an amino acid that cells can synthesize but that also is needed from the diet by stem cells in the skin, to build the keratin protein of hair and outer skin layers. When animals were deprived of serine, skin stem cells devoted less effort to making hair and instead preserved their resources. This makes sense, Fuchs says: Under stressful conditions, when animals aren’t getting enough dietary protein, “what you want is to be able to repair your wounds. You don’t really care so much about making hair.”
This kind of reallocation of resources, other scientists have found, happens on a body-wide level too. When faced with shortages in essential amino acids, or calorie deficits so extreme that the body has a hard time making nonessential ones, liver and muscle tissue is broken down to release amino acids in a bid to preserve a healthy brain. “You don’t destroy your learning, your memory, your behavior, because the assumption is things will get better,” Sabatini says.
But bodies can’t keep this up indefinitely: Decades-old experiments show that mice on diets lacking in certain essential amino acids eventually die from the deficiency. “This is how we know that these amino acids are really essential,” Bröer says.
An herbivore’s dilemma
Some herbivorous animals have evolved smart strategies to prevent such calamities. That’s especially important for sap-feeding insects like aphids, psyllids and white flies — since sap is scarce in amino acids, especially essential ones. So how do the insects get essential amino acids?
Research has revealed that many of them have cultivated symbiotic relationships with bacteria that reside in specialized cells in their abdomens. These bacteria generate most of the essential amino acids and supply them to their hosts. Few aphids live without Buchnera, and no psyllid has been found without Carsonella; mother insects pass them on to their young when the bacteria enter egg cells.
And many sap-feeders don’t stop there. All 36 Hawaiian species of Pariaconus psyllids host the Carsonella bacterium, but many also get essential amino acids that Carsonella doesn’t produce from Makana and Malihini. The precise makeup of these bacterial communities evolved as their insect hosts adopted different diets and thus had different needs, explains entomologist and evolutionary biologist Allison Hansen of the University of California, Riverside.
In a 2024 study, for example, Hansen and her colleagues learned that, when Pariaconus psyllids left the island of Kauai a few million years ago to colonize other Hawaiian islands, some species lost their Malihini bacteria and others lost both Malihini and Makana. These insects had evolved a new habit of stimulating plants to produce thick leaf and stem growths, called galls, to live in and feed on. These galls are rich in amino acids that the psyllids can eat — so, says Hansen, “they started to lose their co-symbionts as they evolved as a gall feeder.”

Insects like psyllids can survive on their protein-poor diets of plant sap because they carry symbiotic bacteria in their bodies that supply essential amino acids. Many psyllids of the Pariaconus genus carry several such bacteria, and these communities have evolved as the insects adapted to different diets. For instance, species like P. dorsotriatus stimulate plants to produce structures called galls (top panel) that enclose the developing insects — and these species have lost some of their symbionts. That’s probably because the galls are nutrient-rich and adequately supply the young insects (bottom panel) with amino acids.
CREDIT: D.M. PERCY
Other herbivores lean on gut-dwelling bacteria to supply amino acids. That includes tree-dwelling turtle ants, which get some nonessential and essential amino acids in this manner, as molecular ecologist Jacob Russell of Drexel University in Pennsylvania and colleagues discovered in 2018. Curiously, although the ants’ diet of nectar, pollen, spores and honeydew probably supplies a full range of amino acids — other insects eating the same do just fine — turtle ants get a big health boost from their gut bacteria. The microbes help to supply the amino acids needed to make the insects’ skin-like cuticle, for example.
The ants likely acquire the bacteria from one another during habitual exchanges of fluid from mouths and anuses. One 2023 study suggests that young queens get bacteria this way before leaving their parental colonies to establish their own. “They’re transmitting those microbes to their offspring,” Russell says.
Some mammals, too, receive a top-up of amino acids from gut bacteria they receive from their mothers during birth and while nursing. Ruminants — herbivores like cows, deer and sheep with microbe-rich four-chambered stomachs — are best known for this. Dairy cows get between 25 and 75 percent of their essential amino acids from gut bacteria, and that is why cows being raised as livestock can be fed on a corn-rich diet that contains little lysine. (Pigs and chickens, which lack such complex digestive systems, require supplements of lysine and other essential amino acids.)
New Mexico’s Newsome suspects that gut bacteria might be helpful protein sources of essential amino acids for many mammals — “I think it’s a process that’s fairly ubiquitous,” he says. Detailed analyses of the blood of herbivorous kangaroo rats of the New Mexico desert, for instance, suggest that more than a third of the essential amino acids in their bodies may originate from gut microbes. And in 2023, a group of scientists in China and the United States identified gut bacteria in giant pandas that seem to modestly boost the amount of essential amino acids in their hosts’ intestines. Perhaps these bacteria help make up for some of the nutritional shortcomings of bamboo, which contains low levels of tryptophan and histidine.
There’s also some evidence that humans may get some essential amino acids from gut microbes, but Bröer says more research is needed to establish how large of a contribution these make to human health. (In any case, he says, the nutrients are still essential; it’s important that vegans and vegetarians get a full range of amino acids from their diet.)
Little by little, science is revealing how animals have managed to survive and thrive, even if they eat only plants, after our ancestors made a risky move to give up the ability to synthesize all amino acids. “Clearly, given that we’re all still here 530 million years later,” Pickard says, “that was a successful strategy.”
10.1146/knowable-120325-1
TAKE A DEEPER DIVE | Explore Related Scholarly Articles