In the biological drama that is a cell’s life, you might think of DNA as the playwright, RNA as the director and proteins as the stars of the show. But life, and living things, are rarely so simple. It turns out that a less understood set of players — a crew of sugar structures known as glycans — gives added nuance and meaning to the action on the cellular stage.
For decades, compared with other biomolecules such as proteins and nucleic acids, these cellular sugar coatings were relatively unexplored. But with the advent of new technologies, including a Nobel-winning analysis technique, scientists are starting to map what’s come to be known as the glycome.
These sugary thickets, they are finding, have big biological consequences. Chemically linked to proteins and lipids on the cell surface, glycans direct communication and the exchange of materials between cells; they guide immune responses and substantially affect a cell’s functions and growth; they’re exploited by disease-causing viruses and other pathogens to gain entry. Glycans attach to biomolecules in every living creature, from single-celled organisms to gigantic blue whales and all the brainy scientists who study how they work.
Like proteins, which are made by linking together individual amino acids, glycans are built from smaller chemical components that shape their function. But unlike DNA, RNA and proteins, whose basic units assemble neatly into linear molecules with predictable properties, glycans are linked to cell-surface proteins and lipids as messier structures with many branches consisting of sugars like glucose, galactose, mannose and xylose.
Glycans are “the face of the cell,” says Laura Kiessling, a chemical biologist at MIT. “They’re how cells can recognize each other.” For example, these cell-surface sugars influence the activity and density of a protein called epidermal growth factor receptor (EGFR) — which is critical for cell growth and proliferation. That, in turn, affects sensitivity to messages from the outside and how quickly the cell divides. Over time, changes in the pattern of glycans can nudge a healthy cell onto a path toward cancer.
And that’s just one cell-surface protein among many. Other glycan interactions can boost anti-tumor immune responses, fight bacterial pathogens and possibly fend off brain aging by maintaining the integrity of the blood-brain barrier.
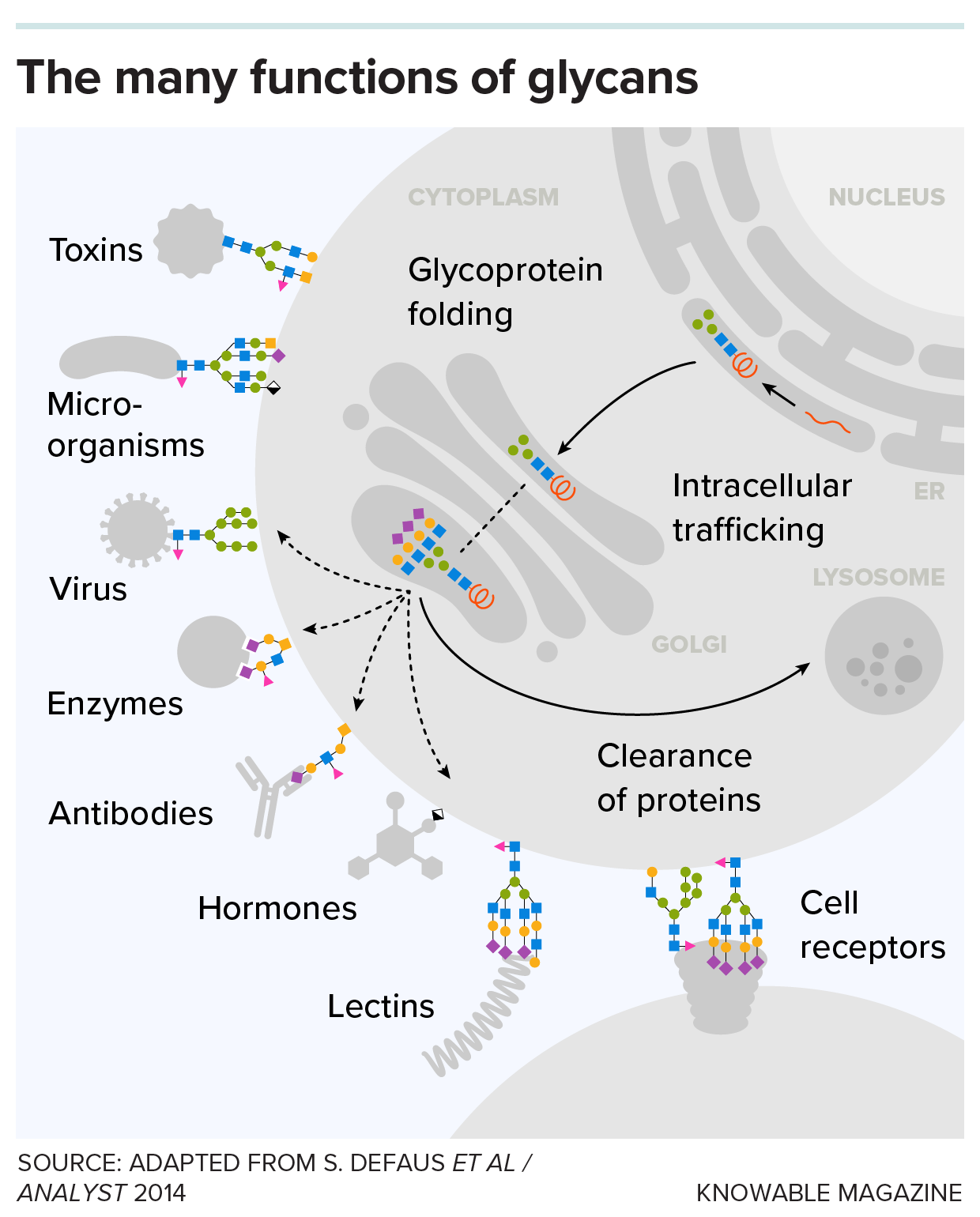
Glycans play myriad roles inside and outside all living cells. When attached to proteins and lipids on the cell's outer membrane, they serve as recognition points for other cells and many molecules, as well as pathogens.
“Glycans can really be a powerful way to drive biology,” says Hans Wandall, a glycobiologist at the University of Copenhagen. A 2024 article in the Annual Review of Biochemistry by Wandall and other researchers catalogs the diverse processes that write the “glycocode” by attaching sugars to cell-surface biomolecules using enzymes, as well as the plethora of glycan-binding proteins that read the code.
A hidden realm
The sheer complexity of all these structures makes it impossible to sequence the glycome — the total set of glycans in an organism — with technologies analogous to the ones routinely used to sequence genetic material and proteins. Plus, glycans don’t assemble into large, complex structures like chromosomes that can be visualized with common lab technologies, says biochemist Richard Cummings, who directs the Harvard Medical School Center for Glycoscience in Boston.
In fact, though scientists had observed glycans by the 1960s, when they saw a fuzzy sugar coating on cells in electron microscope images, it took decades to easily identify specific glycans on the surfaces of living cells. One key technique, which incorporates fluorescent tags on sugars as they are linked to molecules within cells, was recognized with the 2022 Nobel Prize in chemistry.
Researchers are now studying the biological functions of glycomolecules in a more systematic, comprehensive manner, using tools like glycan microarrays, in which biological samples are incubated with slides that contain tiny spots of glycans. Fluorescent probes then measure the interactions of these spots with glycan-binding proteins, antibodies, pathogens or cells.
Boosting immunity, protecting the brain
Cummings and colleagues in Switzerland have used such glycan microarrays to discover that a substantial fraction — more than one-fourth — of the immune system’s most common antibody (immunoglobulin G) binds to sugars and other carbohydrates, not to proteins. That discovery helped to explain why children who lack these particular antibodies are prone to constant bacterial infections. With no way to detect the sugars on bacterial surfaces, their immune systems cannot mount an effective defense against these pathogens, even though they have normal antibody responses to proteins. “That was a breakthrough in understanding the immune system,” says Cummings. “It sees carbohydrates differently from how it sees proteins.”
Kiessling’s lab has used glycan microarrays to study proteins that bind to the mucus that lines the human intestine. One of those proteins, intelectin-2, can bind to both human and bacterial glycans. According to research Kiessling’s MIT team published in January, that’s a one-two punch: Not only can intelectin-2 stabilize the gut’s mucosal barrier by sticking to human glycans, it can also bind glycans on pathogenic bacteria to protect against them. The findings fit well with earlier analyses by Kiessling and other MIT coworkers suggesting a similar dual role for glycans in the mouth’s mucus. “Linking these two functions, which are going to turn out to be general for a lot of these proteins, is really cool,” says Kiessling.
Other research has focused on the glycocalyx — a sugary coating of protein- and lipid-linked glycans that exist on the surface of cells. A 2018 study in mice showed that this layer of glycans is especially thick in the blood-brain barrier, tightly packed cells that regulate what goes in and out of the brain.
The blood-brain barrier breaks down as people age or develop neurodegenerative disease. Sophia Shi, a neurobiologist at Harvard’s Rowland Institute, wondered whether the glycocalyx contributes to that deterioration.
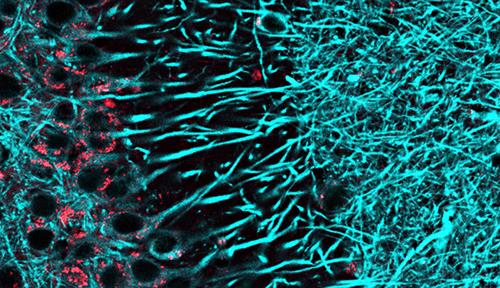
Her hunch was correct. Mentored by Stanford chemist Carolyn Bertozzi and neuroscientist Tony Wyss-Coray, Shi, with colleagues, examined the brains of young and old mice using electron microscopy-based imaging and found that the glycocalyx was unusually thin in the blood vessels of older animals. Shi linked that change to a certain class of sugars called O-linked glycans. In mice lacking the enzymes that assemble these glycans, the blood-brain barrier became leaky. But when Shi and her colleagues restored O-linked glycan production to aging animals using a viral technique, the blood-brain barrier was reinforced.
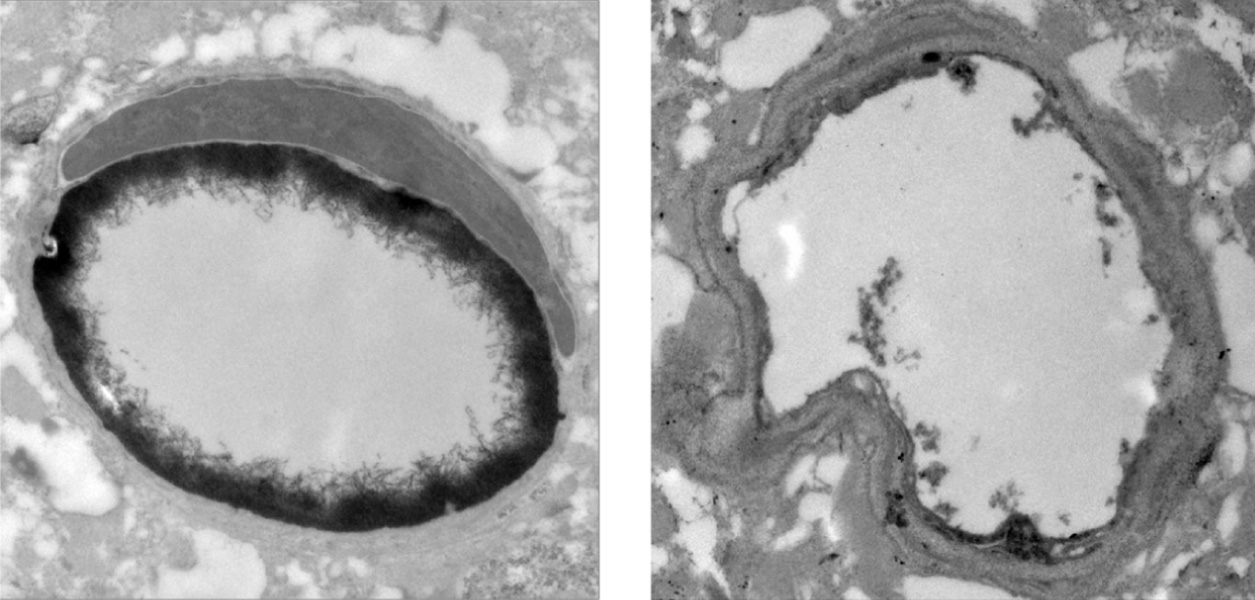
In mice, the blood-brain barrier weakens with age as the layer of glycans on the surface of brain capillaries gets thinner. Left: Capillary from young mouse. Right: capillary from old mouse.
CREDIT: S.M. SHI ET AL / NATURE 2025
These findings are bringing together glycoscience and brain research — two realms that rarely intersect, Shi says. “In glycobiology, people study glycans in a lot of different areas, but not as much in the brain, because it’s a complex organ itself,” Shi notes. After hearing her presentations about the blood-brain barrier glycocalyx, “they’re like, I had no idea that this structure even existed!”
Researchers have only begun to scratch the surface of the glycome’s role in life’s cellular pageant. But it is already clear that these emerging players will have many chances to steal the show as scientists continue to explore the roots of health and disease.